Premalignant states in the skin
The best-defined risk factors for the development of skin cancers in patients with lightly-pigmented skin include immune suppression, aging, and sun exposure. While genetic mutations resulting from ultraviolet light exposure are key events in tumor evolution, epigenetic changes–including altered DNA methylation and histone modifications, which are known features of normal aging–play a less-defined role. This interplay between intrinsic and extrinsic factors that modify cancer risk drives the unifying theme our investigation: defining the molecular drivers of premalignant states in skin cancer.

Characterizing aging phenotypes in murine skin.
We have found the development of canonical, differentially methylated regions of DNA in aging mouse epidermis by whole-genome bisulfite sequencing. These changes are progressive, suggesting a programmed shift in DNA methylation with age. We are actively investigating the mechanisms underlying this change, its functional effects, and whether it modifies cancer susceptibility.
Characterizing epigenetic perturbations that result in premalignant skin phenotypes in humans and mice.
Skin-directed de novo DNA methyltransferase deficiency causes premalignant state in which we identified a focal, canonical DNA hypomethylation phenotype and an expanded proliferative fraction of Dnmt3a-deficient murine epidermal keratinocytes. We are studying the consequences of defined oncogenic drivers in Dnmt3a-deficient skin. We are also premalignant states driven by other genetic and epigenetic perturbations, largely guided by recurrently mutated genes in human skin cancers.

Characterizing genetic and functional changes in human premalignant skin.
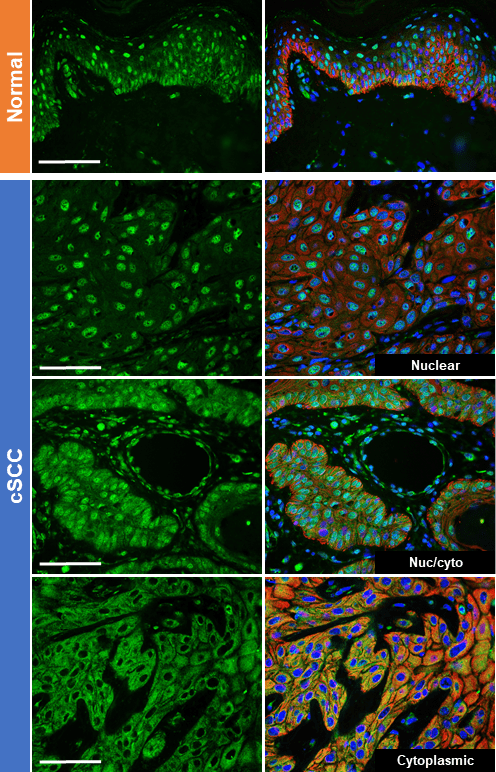
Several premalignant conditions exist in human skin, including actinic keratoses (SCC) and melanocytic nevi (melanoma). We are examining the mechanisms that constrain the transformation of premalignant lesions and how they fail.
Translational studies in skin cancer patients

We have ongoing, collaborative studies examining factors associated with exceptional response to melanoma therapies as well as determining risk of adverse effects associated with melanoma therapies.